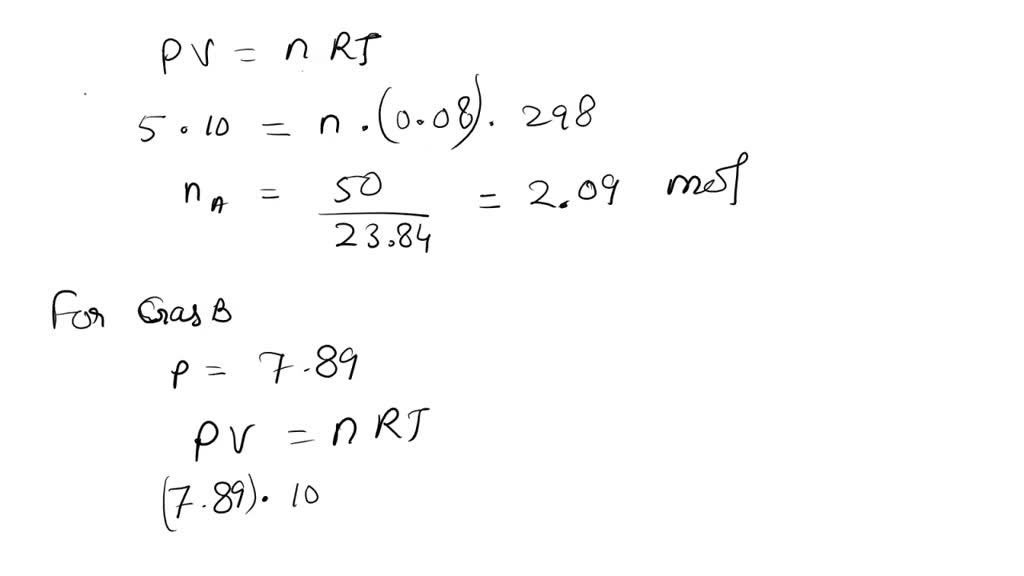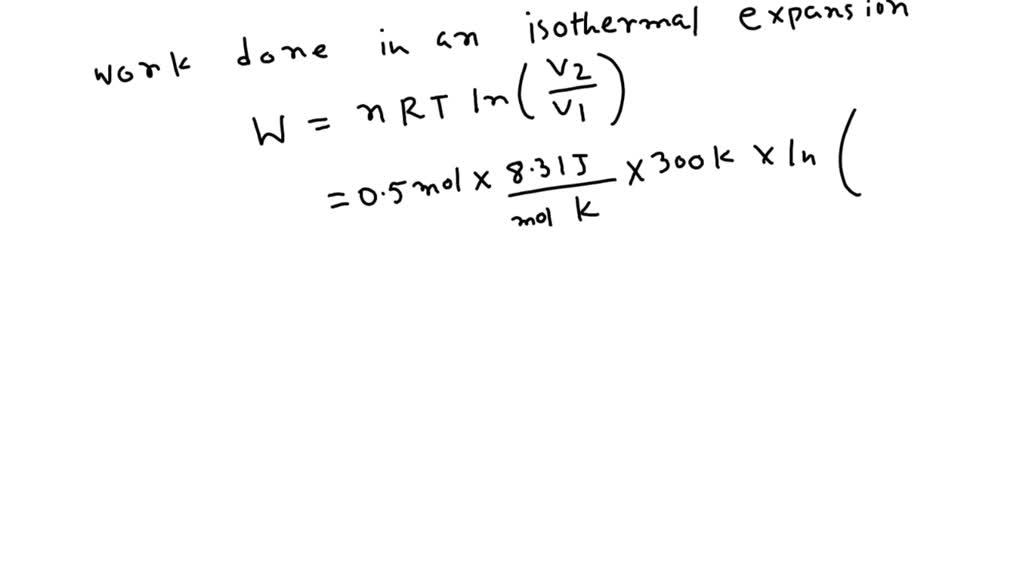
SOLVED: 0.5 mole of gas at temp 300 K expands isothermally from an initial volume of 2.0L to final volume of 6.0L. (a) What is the work done by the gas ? (

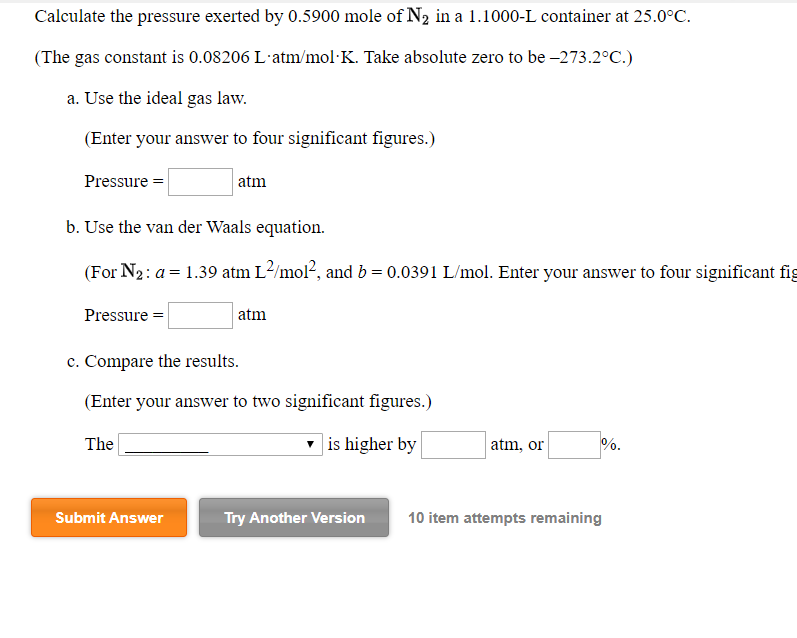
Calculate the temperature of 2 moles of sulphur dioxide gas contained in a 5 L vessel at 10 bar pressure. Given that for SO(2) gas, van der Waals constants are : a=6.7