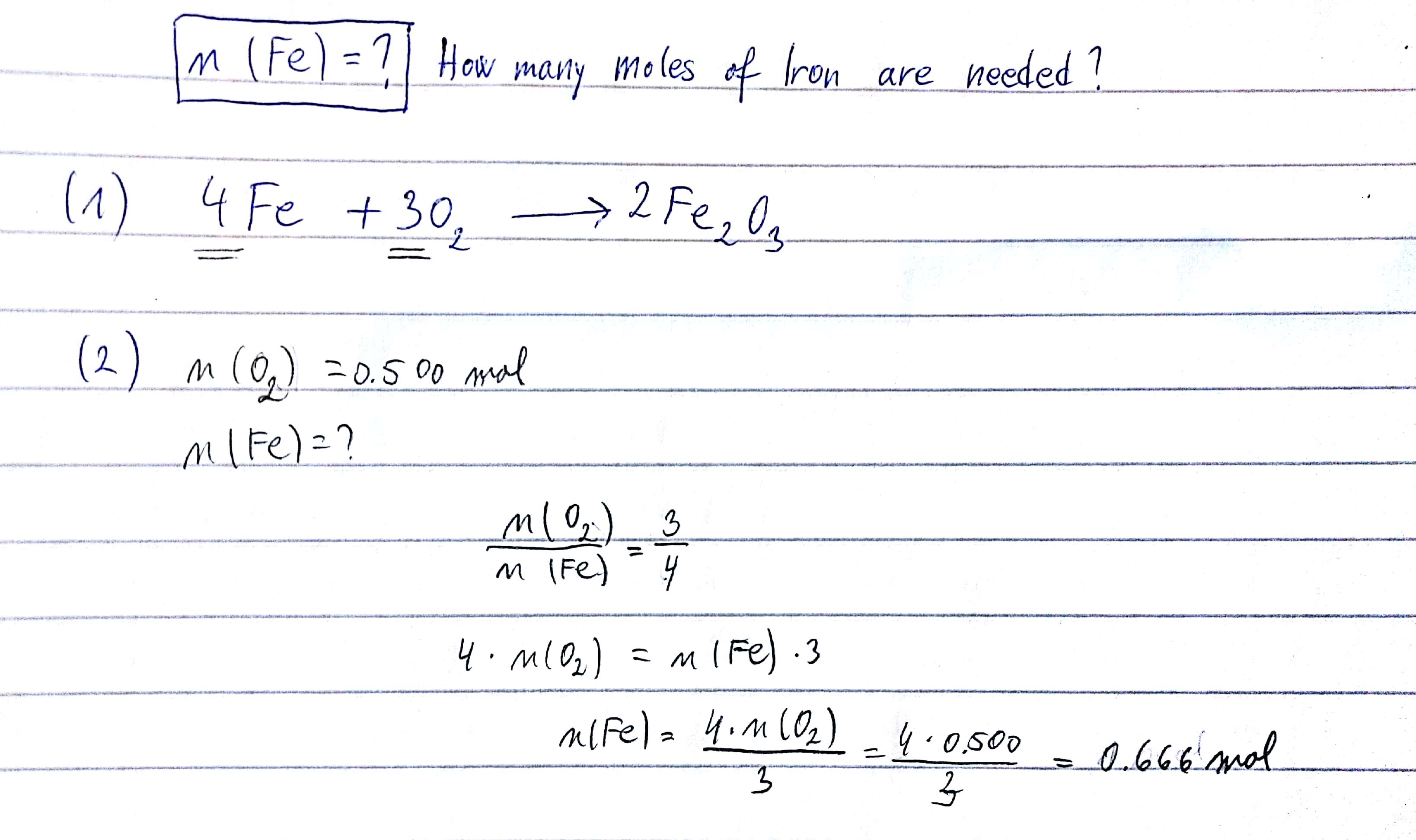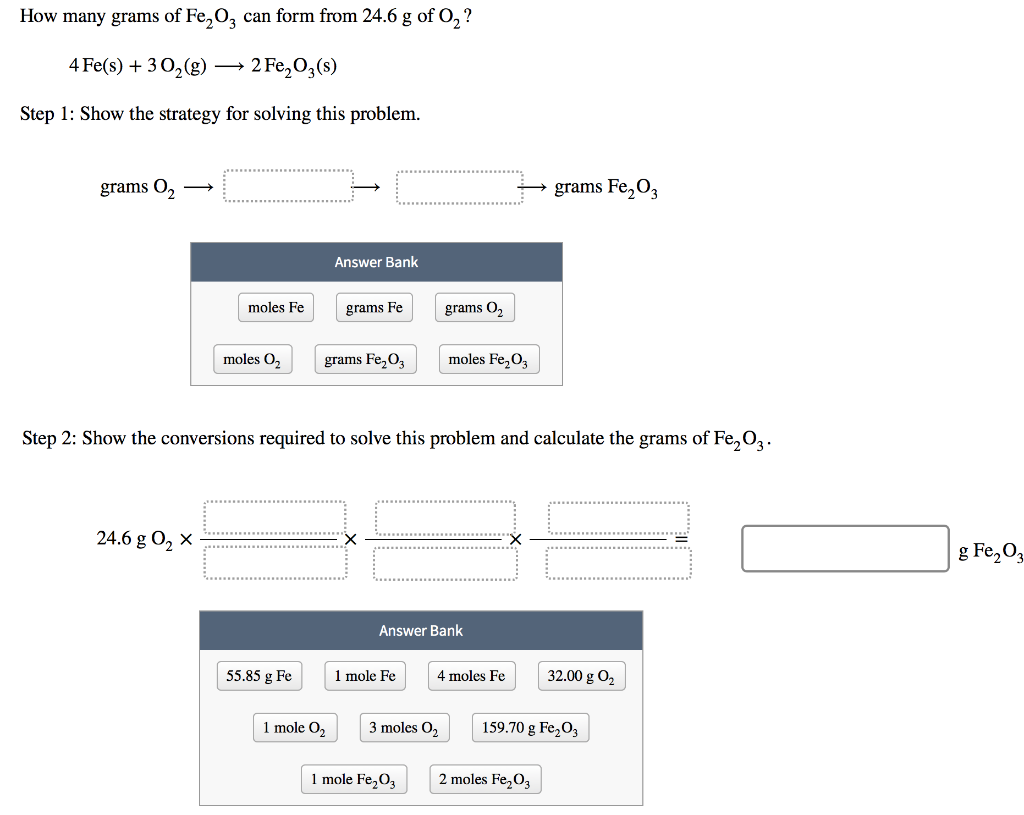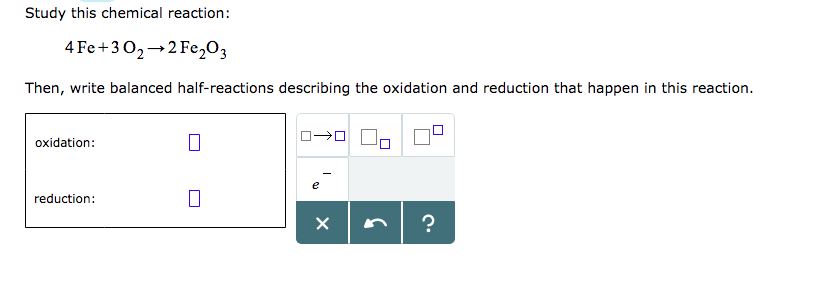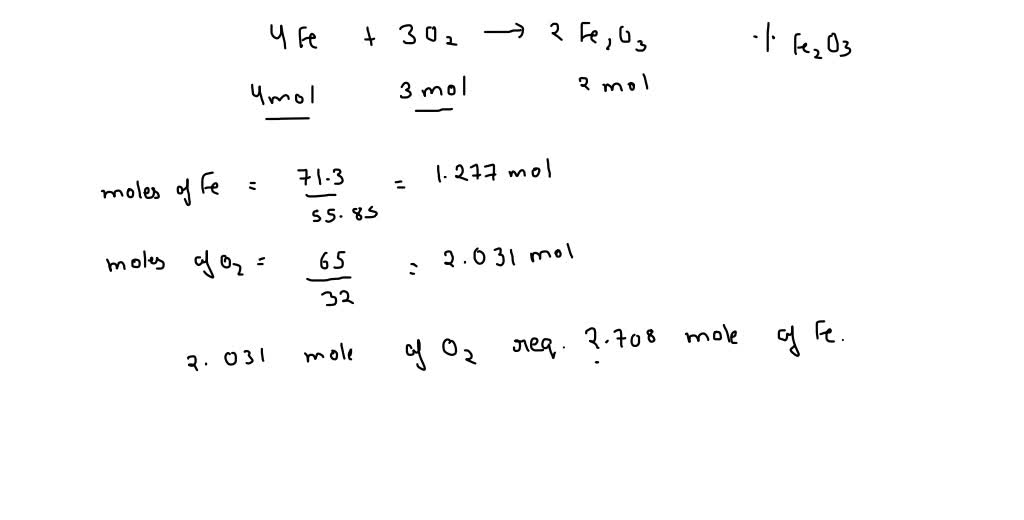
SOLVED: 4Fe(s)+3O2(g)⟶2Fe2O3(s) If 71.3 g of Fe and 65.0 g of O2 are mixed, and 85.0 g of Fe2O3 is actually obtained, what is the percent yield of Fe2O3 for the reaction?

SOLVED: Please provide solution so I can review. Using the reaction: 4Fe + 3O2 → 2Fe2O3 answer the following. How many moles of Fe2O3 are produced when 0.275 moles of Fe is